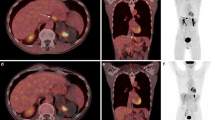
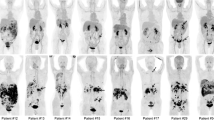
Abstract
Purpose
To identify the biodistribution and diagnostic performance of a novel fibroblast activation protein (FAP) targeted positron emission tomography (PET) tracer, [68Ga]Ga-DOTA-GPFAPI-04, in patients with solid tumors in a head-to-head comparison with [18F]F-FDG.
Methods
Twenty-six patients histologically proven with cancers of nasopharyngeal (n = 5), esophagus (n = 5), gastro-esophagus (n = 1), stomach (n = 7), liver (n = 3), and colorectum (n = 5) were recruited for [68Ga]Ga-DOTA-GPFAPI-04 and [18F]F-FDG PET/CT scans on consecutive days. The primary endpoint was the diagnostic efficacy, with the histological diagnosis and the follow-up results selected as the gold standard. The secondary endpoint was the background uptake pattern. Two experienced nuclear medicine physicians who were blinded to the gold standard results while having essential awareness of the clinical context reviewed the images and labeled lesions by consensus for subsequent software-assisted lesion segmentation. Additionally, background organs were automatically segmented, assisted by artificial intelligence. Volume, mean, and maximum standard uptake values (SUVmean and SUVmax) of all segmentations were recorded. P < 0.05 was deemed as statistically significant.
Results
Significant glandular uptake of [68Ga]Ga-DOTA-GPFAPI-04 was detected in the thyroid, pancreas, and submandibular glands, while moderate uptake was observed in the parotid glands. The myocardium and myometrium exhibited 2–3 times higher uptake of the radiotracer than that of the background levels in blood and liver. A total of 349 targeted lesions, consisting of 324 malignancies and 25 benign lesions, were segmented. [68Ga]Ga-DOTA-GPFAPI-04 is more sensitive than [18F]F-FDG, especially for abdominopelvic dissemination (1.000 vs. 0.475, P < 0.001). Interestingly, [18F]F-FDG demonstrated higher sensitivity for lung metastasis compared to [68Ga]Ga-DOTA-GPFAPI-04 (0.845 vs. 0.682, P = 0.003). The high glandular uptake made it difficult to delineate lesions in close proximity and masked two metastatic lesions in these organs.
Conclusion
Despite prominent glandular uptake, [68Ga]Ga-DOTA-GPFAPI-04 demonstrates favorable diagnostic performance. It is a promising probe scaffold for further development of FAP-targeted tumor theranostic agents.



Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer. 2020;20:174–86. https://doi.org/10.1038/s41568-019-0238-1.
Prive BM, Boussihmad MA, Timmermans B, van Gemert WA, Peters SMB, Derks YHW, et al. Fibroblast activation protein-targeted radionuclide therapy: background, opportunities, and challenges of first (pre)clinical studies. Eur J Nucl Med Mol Imaging. 2023;50:1906–18. https://doi.org/10.1007/s00259-023-06144-0.
Zhang Z, Tao J, Qiu J, Cao Z, Huang H, Xiao J, et al. From basic research to clinical application: targeting fibroblast activation protein for cancer diagnosis and treatment. Cell Oncol (Dordr). 2023. https://doi.org/10.1007/s13402-023-00872-z.
Giesel FL, Kratochwil C, Lindner T, Marschalek MM, Loktev A, Lehnert W, et al. 68Ga-FAPI PET/CT: Biodistribution and preliminary Dosimetry Estimate of 2 DOTA-Containing FAP-Targeting agents in patients with various cancers. J Nucl Med. 2019;60:386–92. https://doi.org/10.2967/jnumed.118.215913.
Kratochwil C, Flechsig P, Lindner T, Abderrahim L, Altmann A, Mier W, et al. 68Ga-FAPI PET/CT: Tracer Uptake in 28 different kinds of Cancer. J Nucl Med. 2019;60:801–5. https://doi.org/10.2967/jnumed.119.227967.
Toms J, Kogler J, Maschauer S, Daniel C, Schmidkonz C, Kuwert T, et al. Targeting fibroblast activation protein: Radiosynthesis and preclinical evaluation of an 18F-Labeled FAP inhibitor. J Nucl Med. 2020;61:1806–13. https://doi.org/10.2967/jnumed.120.242958.
Lai C, Cao R, Li R, He C, Wang X, Shi H, et al. Fibroblast activation protein targeting probe with gly-pro sequence for PET of Glioblastoma. Mol Pharm. 2023;20:4120–8. https://doi.org/10.1021/acs.molpharmaceut.3c00248.
Kersemans K, De Man K, Courtyn J, Van Royen T, Piron S, Moerman L, et al. Automated radiosynthesis of Al[18F]PSMA-11 for large scale routine use. Appl Radiat Isot. 2018;135:19–27. https://doi.org/10.1016/j.apradiso.2018.01.006.
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42:328–54. https://doi.org/10.1007/s00259-014-2961-x.
Wasserthal J, Breit HC, Meyer MT, Pradella M, Hinck D, Sauter AW, et al. TotalSegmentator: robust segmentation of 104 anatomic structures in CT images. Radiol Artif Intell. 2023;5:e230024. https://doi.org/10.1148/ryai.230024.
Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin JC, Pujol S, et al. 3D slicer as an image computing platform for the quantitative Imaging Network. Magn Reson Imaging. 2012;30:1323–41. https://doi.org/10.1016/j.mri.2012.05.001.
Beichel RR, Van Tol M, Ulrich EJ, Bauer C, Chang T, Plichta KA, et al. Semiautomated segmentation of head and neck cancers in 18F-FDG PET scans: a just-enough-interaction approach. Med Phys. 2016;43:2948–64. https://doi.org/10.1118/1.4948679.
Sharma V, McNeill JH. To scale or not to scale: the principles of dose extrapolation. Br J Pharmacol. 2009;157:907–21. https://doi.org/10.1111/j.1476-5381.2009.00267.x.
Demetrius L. Of mice and men. When it comes to studying ageing and the means to slow it down, mice are not just small humans. EMBO Rep. 2005;6:39–44. https://doi.org/10.1038/sj.embor.7400422. Spec No:S.
Wang S, Zhou X, Xu X, Ding J, Liu T, Jiang J, et al. Dynamic PET/CT imaging of 68Ga-FAPI-04 in Chinese subjects. Front Oncol. 2021;11:651005. https://doi.org/10.3389/fonc.2021.651005.
Kumar S, Borish K, Dey S, Nagesh J, Das A. Sequence dependent folding motifs of the secondary structures of gly-pro and Pro-gly containing oligopeptides. Phys Chem Chem Phys. 2022;24:18408–18. https://doi.org/10.1039/d2cp01306a.
Minelli A, Conte C, Cacciatore I, Cornacchia C, Pinnen F. Molecular mechanism underlying the cerebral effect of gly-pro-glu tripeptide bound to L-dopa in a Parkinson’s animal model. Amino Acids. 2012;43:1359–67. https://doi.org/10.1007/s00726-011-1210-x.
Ding H, Liang J, Qiu L, Xu T, Cai L, Wan Q, et al. Prospective comparison of 68Ga-FAPI-04 and 18F-FDG PET/CT for tumor staging in nasopharyngeal carcinoma. Front Oncol. 2022;12:1047010. https://doi.org/10.3389/fonc.2022.1047010.
Qin C, Liu F, Huang J, Ruan W, Liu Q, Gai Y, et al. A head-to-head comparison of 68Ga-DOTA-FAPI-04 and 18F-FDG PET/MR in patients with nasopharyngeal carcinoma: a prospective study. Eur J Nucl Med Mol Imaging. 2021;48:3228–37. https://doi.org/10.1007/s00259-021-05255-w.
Wang L, Tang G, Hu K, Liu X, Zhou W, Li H, et al. Comparison of 68Ga-FAPI and 18F-FDG PET/CT in the evaluation of Advanced Lung Cancer. Radiology. 2022;303:191–9. https://doi.org/10.1148/radiol.211424.
Li M, Younis MH, Zhang Y, Cai W, Lan X. Clinical summary of fibroblast activation protein inhibitor-based radiopharmaceuticals: cancer and beyond. Eur J Nucl Med Mol Imaging. 2022;49:2844–68. https://doi.org/10.1007/s00259-022-05706-y.
Chen R, Yang X, Yu X, Zhou X, Ng YL, Zhao H, et al. Tumor-to-blood ratio for assessment of fibroblast activation protein receptor density in pancreatic cancer using 68Ga-FAPI-04. Eur J Nucl Med Mol Imaging. 2023;50:929–36. https://doi.org/10.1007/s00259-022-06010-5.
Wang L, Wang Y, Wang J, Xiao M, Xi XY, Chen BX, et al. Myocardial activity at 18F-FAPI PET/CT and risk for Sudden Cardiac Death in Hypertrophic Cardiomyopathy. Radiology. 2023;306:e221052. https://doi.org/10.1148/radiol.221052.
Zhang X, Song W, Qin C, Song Y, Liu F, Hu F, et al. Uterine uptake of 68Ga-FAPI-04 in Uterine Pathology and Physiology. Clin Nucl Med. 2022;47:7–13. https://doi.org/10.1097/RLU.0000000000003968.
Nakamoto Y, Baba S, Kaida H, Manabe O, Uehara T. Recent topics in fibroblast activation protein inhibitor-PET/CT: clinical and pharmacological aspects. Ann Nucl Med. 2024;38:10–9. https://doi.org/10.1007/s12149-023-01873-6.
Pang Y, Zhao L, Luo Z, Hao B, Wu H, Lin Q, et al. Comparison of 68Ga-FAPI and 18F-FDG uptake in gastric, duodenal, and colorectal cancers. Radiology. 2021;298:393–402. https://doi.org/10.1148/radiol.2020203275.
Cai J, Yang D, Sun H, Xiao L, Han F, Zhang M, et al. A multifactorial analysis of FAP to regulate gastrointestinal cancers progression. Front Immunol. 2023;14:1183440. https://doi.org/10.3389/fimmu.2023.1183440.
Zhao L, Pang Y, Luo Z, Fu K, Yang T, Zhao L, et al. Role of 68Ga-DOTA-FAPI-04 PET/CT in the evaluation of peritoneal carcinomatosis and comparison with 18F-FDG PET/CT. Eur J Nucl Med Mol Imaging. 2021;48:1944–55. https://doi.org/10.1007/s00259-020-05146-6.
Pure E, Blomberg R. Pro-tumorigenic roles of fibroblast activation protein in cancer: back to the basics. Oncogene. 2018;37:4343–57. https://doi.org/10.1038/s41388-018-0275-3.
Braune A, Oehme L, Freudenberg R, Hofheinz F, van den Hoff J, Kotzerke J, et al. Comparison of image quality and spatial resolution between 18F, 68Ga, and 64Cu phantom measurements using a digital Biograph Vision PET/CT. EJNMMI Phys. 2022;9:58. https://doi.org/10.1186/s40658-022-00487-7.
Funding
This work was supported by the fund from the National Natural Science Foundation of China (81971645 and U2267221), Guangdong Provincial People’s Hospital (KY0120211130), Guangdong Provincial Key Laboratory of Artificial Intelligence in Medical Image Analysis and Application (2022B1212010011), Shanghai Municipal Science and Technology Major Project (TM202301H003), Gansu Science and Technology Major Project (23ZDFA014), and Shandong Laboratory Program (SYS202205).
Author information
Authors and Affiliations
Contributions
Lei Jiang and Zhen Cheng designed the study, Hui Yuan, Entao Liu, Qing Zhang, and Yuxiang Shang collected PET/CT data, Guojin Zhang and Chaoquan Lai synthesized the tracer, Hui Yuan and Entao Liu analyzed the data and carried out the statistical analysis, and all authors wrote and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
This is a prospective study (NCT06186427), locally approved by the Ethics Committee of Guangdong Provincial People’s Hospital (No. XJS2023-005-01) and conducted following the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to participate
All subjects gave written consent before participating in the study.
Consent for publication
The authors affirm that all human participants involved in this study have signed the consent form and agreed to the publication of their medical imaging and records in an anonymized format for the purposes of scientific research.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuan, H., Liu, E., Zhang, G. et al. Diagnostic efficacy of [68Ga]Ga-DOTA-GPFAPI-04 in patients with solid tumors in a head-to-head comparison with [18F]F-FDG: results from a prospective clinical study. Eur J Nucl Med Mol Imaging (2024). https://doi.org/10.1007/s00259-024-06756-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00259-024-06756-0