Abstract
Purpose
Biochemical recurrence (BCR) following radical prostatectomy (RP) is a significant concern for patients with prostate cancer. Reliable prediction models are needed to identify patients at risk for BCR and facilitate appropriate management. This study aimed to develop and validate a clinical-radiomics model based on preoperative [18 F]PSMA-1007 PET for predicting BCR-free survival (BRFS) in patients who underwent RP for prostate cancer.
Materials and methods
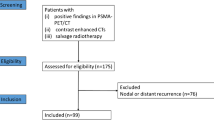
A total of 236 patients with histologically confirmed prostate cancer who underwent RP were retrospectively analyzed. All patients had a preoperative [18 F]PSMA-1007 PET/CT scan. Radiomics features were extracted from the primary tumor region on PET images. A radiomics signature was developed using the least absolute shrinkage and selection operator (LASSO) Cox regression model. The performance of the radiomics signature in predicting BRFS was assessed using Harrell’s concordance index (C-index). The clinical-radiomics nomogram was constructed using the radiomics signature and clinical features. The model was externally validated in an independent cohort of 98 patients.
Results
The radiomics signature comprised three features and demonstrated a C-index of 0.76 (95% CI: 0.60–0.91) in the training cohort and 0.71 (95% CI: 0.63–0.79) in the validation cohort. The radiomics signature remained an independent predictor of BRFS in multivariable analysis (HR: 2.48, 95% CI: 1.47–4.17, p < 0.001). The clinical-radiomics nomogram significantly improved the prediction performance (C-index: 0.81, 95% CI: 0.66–0.95, p = 0.007) in the training cohort and (C-index: 0.78 95% CI: 0.63–0.89, p < 0.001) in the validation cohort.
Conclusion
We developed and validated a novel [18 F]PSMA-1007 PET-based clinical-radiomics model that can predict BRFS following RP in prostate cancer patients. This model may be useful in identifying patients with a higher risk of BCR, thus enabling personalized risk stratification and tailored management strategies.








Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
24 May 2024
A Correction to this paper has been published: https://doi.org/10.1007/s00259-024-06752-4
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and Mortality Worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. https://doi.org/10.3322/caac.21660.
Shao N, Wang Y, Jiang WY, Qiao D, Zhang SG, Wu Y, et al. Immunotherapy and endothelin receptor antagonists for treatment of castration-resistant prostate cancer. Int J Cancer. 2013;133:1743–50. https://doi.org/10.1002/ijc.28162.
Cornford P, Bellmunt J, Bolla M, Briers E, De Santis M, Gross T, et al. EAU-ESTRO-SIOG guidelines on prostate Cancer. Part II: treatment of relapsing, metastatic, and castration-resistant prostate Cancer. Eur Urol. 2017;71:630–42. https://doi.org/10.1016/j.eururo.2016.08.002.
Rodrigues G, Warde P, Pickles T, Crook J, Brundage M, Souhami L, et al. Pre-treatment risk stratification of prostate cancer patients: a critical review. Can Urol Assoc J. 2012;6:121–7. https://doi.org/10.5489/cuaj.11085.
Mesko S, Marks L, Ragab O, Patel S, Margolis DA, Demanes DJ, et al. Targeted prostate biopsy gleason score heterogeneity and implications for risk stratification. Am J Clin Oncol. 2018;41:497–501. https://doi.org/10.1097/COC.0000000000000308.
Kasperzyk JL, Finn SP, Flavin R, Fiorentino M, Lis R, Hendrickson WK, et al. Prostate-specific membrane antigen protein expression in tumor tissue and risk of lethal prostate cancer. Cancer Epidemiol Biomarkers Prev. 2013;22:2354–63. https://doi.org/10.1158/1055-9965.EPI-13-0668.
Jones W, Griffiths K, Barata PC, Paller CJ. PSMA Theranostics: review of the current status of PSMA-Targeted imaging and Radioligand Therapy. Cancers (Basel). 2020;12. https://doi.org/10.3390/cancers12061367.
Mokoala K, Lawal I, Lengana T, Kgatle M, Giesel FL, Vorster M, et al. PSMA Theranostics: Sci Pract Cancers (Basel). 2021;13. https://doi.org/10.3390/cancers13153904.
Fendler WP, Calais J, Allen-Auerbach M, Bluemel C, Eberhardt N, Emmett L, et al. (68)Ga-PSMA-11 PET/CT interobserver agreement for prostate Cancer assessments: an International Multicenter prospective study. Journal of nuclear medicine: official publication. Soc Nuclear Med. 2017;58:1617–23. https://doi.org/10.2967/jnumed.117.190827.
Duan H, Baratto L, Hatami N, Liang T, Mari Aparici C, Davidzon GA, et al. 68Ga-PSMA11 PET/CT for biochemically recurrent prostate cancer: influence of dual-time and PMT- vs SiPM-based detectors. Translational Oncol. 2022;15:101293. https://doi.org/10.1016/j.tranon.2021.101293.
Mattiolli AB, Santos A, Vicente A, Queiroz M, Bastos D, Herchenhorn D, et al. Impact of 68GA-PSMA PET / CT on treatment of patients with recurrent / metastatic high risk prostate cancer - a multicenter study. Int Braz J Urol. 2018;44:892–9. https://doi.org/10.1590/S1677-5538.IBJU.2017.0632.
Dekalo S, Kuten J, Mintz I, Fahoum I, Gitstein G, Keizman D, et al. Preoperative 68Ga-PSMA PET/CT defines a subgroup of high-risk prostate cancer patients with favorable outcomes after radical prostatectomy and lymph node dissection. Prostate Cancer Prostatic Dis. 2021;24:910–6. https://doi.org/10.1038/s41391-021-00347-y.
Draulans C, De Roover R, van der Heide UA, Kerkmeijer L, Smeenk RJ, Pos F, et al. Optimal (68)Ga-PSMA and (18)F-PSMA PET window levelling for gross tumour volume delineation in primary prostate cancer. Eur J Nucl Med Mol Imaging. 2021;48:1211–8. https://doi.org/10.1007/s00259-020-05059-4.
Hong JJ, Liu BL, Wang ZQ, Tang K, Ji XW, Yin WW, et al. The value of (18)F-PSMA-1007 PET/CT in identifying non-metastatic high-risk prostate cancer. EJNMMI Res. 2020;10:138. https://doi.org/10.1186/s13550-020-00730-1.
Giesel FL, Knorr K, Spohn F, Will L, Maurer T, Flechsig P, et al. Detection efficacy of (18)F-PSMA-1007 PET/CT in 251 patients with biochemical recurrence of prostate Cancer after Radical Prostatectomy. Journal of nuclear medicine: official publication. Soc Nuclear Med. 2019;60:362–8. https://doi.org/10.2967/jnumed.118.212233.
Zhou X, Jiang X, Liu L, Wang X, Li C, Yao Y, et al. Evaluation of (18)F-PSMA-1007 PET/CT in prostate cancer patients with biochemical recurrence after radical prostatectomy. Transl Oncol. 2022;15:101292. https://doi.org/10.1016/j.tranon.2021.101292.
Lambin P, Leijenaar RTH, Deist TM, Peerlings J, de Jong EEC, van Timmeren J, et al. Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol. 2017;14:749–62. https://doi.org/10.1038/nrclinonc.2017.141.
Gillies RJ, Kinahan PE, Hricak H. Radiomics: images are more than pictures, they are data. Radiology. 2016;278:563–77. https://doi.org/10.1148/radiol.2015151169.
Xue XQ, Yu WJ, Shi X, Shao XL, Wang YT. (18)F-FDG PET/CT-based radiomics nomogram for the preoperative prediction of lymph node metastasis in gastric cancer. Front Oncol. 2022;12:911168. https://doi.org/10.3389/fonc.2022.911168.
Wang L, Li T, Hong J, Zhang M, Ouyang M, Zheng X, et al. (18)F-FDG PET-based radiomics model for predicting occult lymph node metastasis in clinical N0 solid lung adenocarcinoma. Quant Imaging Med Surg. 2021;11:215–25. https://doi.org/10.21037/qims-20-337.
Xue B, Wu S, Zhang M, Hong J, Liu B, Xu N, et al. A radiomic-based model of different contrast-enhanced CT phase for differentiate intrahepatic cholangiocarcinoma from inflammatory mass with hepatolithiasis. Abdom Radiol (NY). 2021;46:3835–44. https://doi.org/10.1007/s00261-021-03027-6.
Cardinale J, Martin R, Remde Y, Schafer M, Hienzsch A, Hubner S, et al. Procedures for the GMP-Compliant production and Quality Control of [(18)F]PSMA-1007: a Next Generation Radiofluorinated Tracer for the detection of prostate Cancer. Pharmaceuticals (Basel). 2017;10. https://doi.org/10.3390/ph10040077.
Mottet N, van den Bergh RCN, Briers E, Van den Broeck T, Cumberbatch MG, De Santis M, et al. EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate Cancer-2020 update. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2021;79:243–62. https://doi.org/10.1016/j.eururo.2020.09.042.
Mazzone E, Gandaglia G, Ploussard G, Marra G, Valerio M, Campi R, et al. Risk stratification of patients candidate to Radical Prostatectomy based on clinical and multiparametric magnetic resonance imaging parameters: Development and External Validation of Novel Risk groups. Eur Urol. 2022;81:193–203. https://doi.org/10.1016/j.eururo.2021.07.027.
Roberts MJ, Morton A, Papa N, Franklin A, Raveenthiran S, Yaxley WJ, et al. Primary tumour PSMA intensity is an independent prognostic biomarker for biochemical recurrence-free survival following radical prostatectomy. Eur J Nucl Med Mol Imaging. 2022;49:3289–94. https://doi.org/10.1007/s00259-022-05756-2.
Lambin P, Rios-Velazquez E, Leijenaar R, Carvalho S, van Stiphout RG, Granton P et al. Radiomics: extracting more information from medical images using advanced feature analysis. European journal of cancer (Oxford, England: 1990). 2012;48:441-6. https://doi.org/10.1016/j.ejca.2011.11.036.
Jia TY, Xiong JF, Li XY, Yu W, Xu ZY, Cai XW, et al. Identifying EGFR mutations in lung adenocarcinoma by noninvasive imaging using radiomics features and random forest modeling. Eur Radiol. 2019;29:4742–50. https://doi.org/10.1007/s00330-019-06024-y.
Thawani R, McLane M, Beig N, Ghose S, Prasanna P, Velcheti V, et al. Radiomics and radiogenomics in lung cancer: a review for the clinician. Lung Cancer. 2018;115:34–41. https://doi.org/10.1016/j.lungcan.2017.10.015.
Lee G, Lee HY, Park H, Schiebler ML, van Beek EJR, Ohno Y, et al. Radiomics and its emerging role in lung cancer research, imaging biomarkers and clinical management: state of the art. Eur J Radiol. 2017;86:297–307. https://doi.org/10.1016/j.ejrad.2016.09.005.
Feng ST, Jia Y, Liao B, Huang B, Zhou Q, Li X, et al. Preoperative prediction of microvascular invasion in hepatocellular cancer: a radiomics model using Gd-EOB-DTPA-enhanced MRI. Eur Radiol. 2019;29:4648–59. https://doi.org/10.1007/s00330-018-5935-8.
Li H, Mendel KR, Lan L, Sheth D, Giger ML. Digital mammography in breast Cancer: Additive Value of Radiomics of breast parenchyma. Radiology. 2019;291:15–20. https://doi.org/10.1148/radiol.2019181113.
Zang S, Ai S, Yang R, Zhang P, Wu W, Zhao Z, et al. Development and validation of (68)Ga-PSMA-11 PET/CT-based radiomics model to detect primary prostate cancer. EJNMMI Res. 2022;12:63. https://doi.org/10.1186/s13550-022-00936-5.
Spohn SKB, Schmidt-Hegemann NS, Ruf J, Mix M, Benndorf M, Bamberg F, et al. Development of PSMA-PET-guided CT-based radiomics signature to predict biochemical recurrence after salvage radiotherapy. Eur J Nucl Med Mol Imaging. 2023. https://doi.org/10.1007/s00259-023-06195-3.
Chen A, Lu L, Pu X, Yu T, Yang H, Schwartz LH, et al. CT-Based Radiomics Model for Predicting Brain Metastasis in Category T1 lung adenocarcinoma. AJR Am J Roentgenol. 2019;213:134–9. https://doi.org/10.2214/AJR.18.20591.
Ji GW, Zhu FP, Zhang YD, Liu XS, Wu FY, Wang K, et al. A radiomics approach to predict lymph node metastasis and clinical outcome of intrahepatic cholangiocarcinoma. Eur Radiol. 2019;29:3725–35. https://doi.org/10.1007/s00330-019-06142-7.
Tan Y, Zhang ST, Wei JW, Dong D, Wang XC, Yang GQ, et al. A radiomics nomogram may improve the prediction of IDH genotype for astrocytoma before surgery. Eur Radiol. 2019;29:3325–37. https://doi.org/10.1007/s00330-019-06056-4.
Zhang R, Xu L, Wen X, Zhang J, Yang P, Zhang L, et al. A nomogram based on bi-regional radiomics features from multimodal magnetic resonance imaging for preoperative prediction of microvascular invasion in hepatocellular carcinoma. Quant Imaging Med Surg. 2019;9:1503–15. https://doi.org/10.21037/qims.2019.09.07.
Tsurumaki Sato Y, Fukuhara H, Suzuki M, Fujimura T, Nakagawa T, Nishimatsu H, et al. Long-term results of radical prostatectomy with immediate adjuvant androgen deprivation therapy for pT3N0 prostate cancer. BMC Urol. 2014;14:13. https://doi.org/10.1186/1471-2490-14-13.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Tiancheng Li: data acquisition, literature research, and manuscript writing. Mimi Xu, Yinuo Liu, Guolin Wang and Kaifeng Liu: data acquisition and review, Shuye Yang, Kui Zhao and Xinhui Su: study design and theoretical support. Tiancheng Li and Xinhui Su: design of the research program, review and revision of the manuscript. All the authors agreed on the content of the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Ethics Committee of the First Affiliated Hospital, College of Medicine, Zhejiang University.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
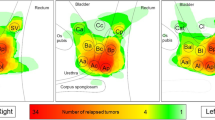
The original online version of this article was revised: The authors regret that the version of Figure 7 that appears in the published original article is incorrect. Both the correct and incorrect Figure 7 are provided in the erratum article.
The original article has been corrected.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, T., Xu, M., Yang, S. et al. Development and validation of [18 F]-PSMA-1007 PET-based radiomics model to predict biochemical recurrence-free survival following radical prostatectomy. Eur J Nucl Med Mol Imaging (2024). https://doi.org/10.1007/s00259-024-06734-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00259-024-06734-6